I almost titled this post Daily Mail celebrates work of immigrants shocker but as they have written a pretty accurate article on work I am part of, that would be a bit ungrateful. Yesterday a paper came out in Physical Review Letters that I am really rather proud of, although I made only a small contribution to it. Most of the credit should go to Andrea Fortini, who discovered the effect the paper describes, and to Nacho Martin-Fabiani and Joe Keddie who did the experiments to show that it works in the real world too. Andrea found the effect in computer simulations. We also had help from collaborators in Lyon who made the particles Nacho used.
Reducing the risk of heart disease with the aid of Russian Roulette
It is almost always easier to borrow ideas and techniques from other fields than to reinvent them. A PhD student, another academic and I, are studying two competing processes. These are crystallisation into two different crystals, called alpha and gamma, of a small molecule called glycine. The formation of alpha and gamma appear to be mutually exclusive, one or the other forms, not both. Crystallisation is statistical, it is at least partly random, and they are irreversible, once a crystal forms it persists.
One hundred and twenty five billion reasons to care about crystallisation
 Lipitor is a cholesterol-lowering drug sold by the pharma multinational Pfizer. People who want to lower their cholesterol take it every day for years or decades. High cholesterol is a common problem, bacon and cakes are just too tasty, and so the market for drugs like lipitor is absolutely enormous. It has been estimated that over fifteen years, Pfizer made $125 billion from lipitor. That’s a lot of money. I don’t have the figures but presumably a billion plus of that was in the UK (i.e., paid for by the NHS). So if you are a taxpayer even if you haven’t taken it, you’ve paid for it. So I’ve paid for it, but I am also interested in lipitor as I study crystallisation, and like most drugs lipitor tablets contain the drug in crystalline form.
Lipitor is a cholesterol-lowering drug sold by the pharma multinational Pfizer. People who want to lower their cholesterol take it every day for years or decades. High cholesterol is a common problem, bacon and cakes are just too tasty, and so the market for drugs like lipitor is absolutely enormous. It has been estimated that over fifteen years, Pfizer made $125 billion from lipitor. That’s a lot of money. I don’t have the figures but presumably a billion plus of that was in the UK (i.e., paid for by the NHS). So if you are a taxpayer even if you haven’t taken it, you’ve paid for it. So I’ve paid for it, but I am also interested in lipitor as I study crystallisation, and like most drugs lipitor tablets contain the drug in crystalline form.
Iron & Steel
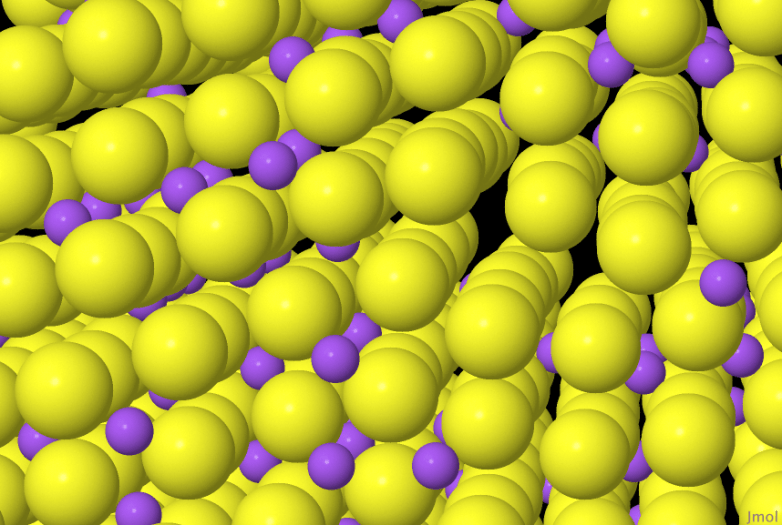 Steel is not pure iron, it contains a small fraction of carbon that transforms the soft pure iron into the much tougher steel. I guess I have known that for a long time. But I have not really thought about how the carbon is incorporated into the crystalline iron. Dissolving salt in liquid water is straightforward. The ions of sodium and chloride just diffuse around in the liquid surrounded by the diffusing molecules of water. This is a liquid solution, a solution of salt in liquid water. Steel is a solid solution, it is carbon dissolved in solid iron.
Steel is not pure iron, it contains a small fraction of carbon that transforms the soft pure iron into the much tougher steel. I guess I have known that for a long time. But I have not really thought about how the carbon is incorporated into the crystalline iron. Dissolving salt in liquid water is straightforward. The ions of sodium and chloride just diffuse around in the liquid surrounded by the diffusing molecules of water. This is a liquid solution, a solution of salt in liquid water. Steel is a solid solution, it is carbon dissolved in solid iron.
The economics of life, death and weekend working
The Health Secretary Jeremy Hunt has been going round saying* “At the moment, for example, if you have a stroke at the weekend you are 20% more likely to die. That cannot be right, and that is something every doctor wants to sort out as well.”. This is a rather alarming way of saying that 11.1% of those admitted on a weekday because they have had a stroke are dead within 30 days, whereas 12.9% of those admitted on a weekend die within 30 days. This is from work of Roberts and coworkers.
Tiny tiny origami
 My Christmas reading has included a PhD* thesis, I am external examiner for a student at Oxford, and the viva is mid-January. The thesis is on the computer simulations of a model of DNA. The simulations are of what is called DNA origami. Origami is of course folding up a sheet of paper in a precise way, to make a paper plane, paper flower, etc.
My Christmas reading has included a PhD* thesis, I am external examiner for a student at Oxford, and the viva is mid-January. The thesis is on the computer simulations of a model of DNA. The simulations are of what is called DNA origami. Origami is of course folding up a sheet of paper in a precise way, to make a paper plane, paper flower, etc.
Interwoven networks of crystals
Jelly is bad for our nerves
My Christmas viewing has included (amongst the Strictly, Agatha Christie adaptation, etc) a webinar entitled Fluid Business: Could “Liquid”Protein Herald Neurodegeneration? The webinar is on droplet-like structures inside nerve cells that may be associated with some diseases that kill these nerve cells, such as Alzheimer’s, amyotrophic lateral sclerosis (ALS), etc. The webinar includes short talks by a number of scientists, including a presentation by Peter St George-Hyslop that is based on a very recent paper in Neuron. The report reports a lot of work by a small army of scientists on a protein called FUS. Some mutant variants of FUS are associated with the disease ALS.
Teaching till I drop, and now some chaos
 Most of my teaching is in the semester whose teaching weeks ended yesterday — it has been a bit crazy. I was more-or-less completely revising two of the three courses I was teaching, so I was a like a little hamster perpetually running on a hamster wheel of lecture and question sheet writing.
Most of my teaching is in the semester whose teaching weeks ended yesterday — it has been a bit crazy. I was more-or-less completely revising two of the three courses I was teaching, so I was a like a little hamster perpetually running on a hamster wheel of lecture and question sheet writing.
Many genes, simple models
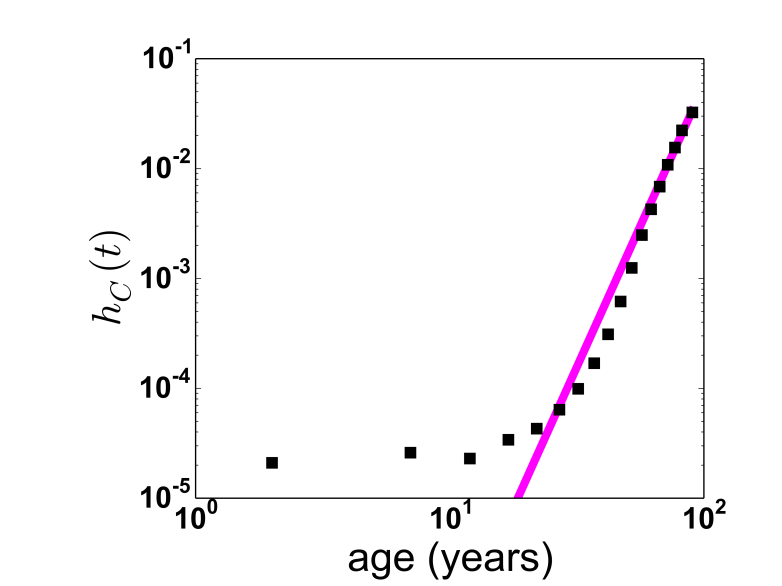
Next week is the last teaching week of this semester. I have been revising almost from scratch both of my second year courses, which at times has taken most of my waking moments. Most of my teaching is this semester, so I have been crazy busy. But I have had time to add a small new bit to my final-year biological physics course. This is on the fact that many aspects of our bodies, and the diseases that afflict them, are controlled not by a single gene, but by many.