 Steel is not pure iron, it contains a small fraction of carbon that transforms the soft pure iron into the much tougher steel. I guess I have known that for a long time. But I have not really thought about how the carbon is incorporated into the crystalline iron. Dissolving salt in liquid water is straightforward. The ions of sodium and chloride just diffuse around in the liquid surrounded by the diffusing molecules of water. This is a liquid solution, a solution of salt in liquid water. Steel is a solid solution, it is carbon dissolved in solid iron.
Steel is not pure iron, it contains a small fraction of carbon that transforms the soft pure iron into the much tougher steel. I guess I have known that for a long time. But I have not really thought about how the carbon is incorporated into the crystalline iron. Dissolving salt in liquid water is straightforward. The ions of sodium and chloride just diffuse around in the liquid surrounded by the diffusing molecules of water. This is a liquid solution, a solution of salt in liquid water. Steel is a solid solution, it is carbon dissolved in solid iron.
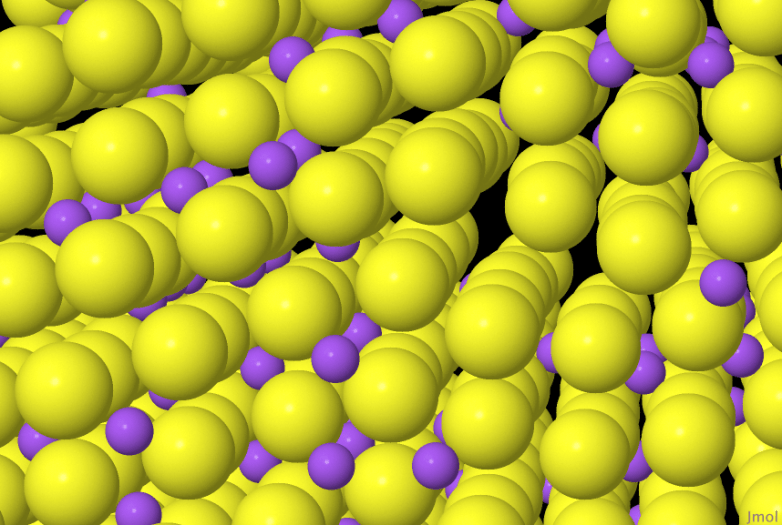
But iron is a crystal, and so the iron atoms are arranged in a regular repeating lattice with only small gaps between them, so how do the carbon atoms fit in? The answer is that carbon atoms are rather smaller than iron atoms so they can squeeze into these gaps.
I learnt all this after I had made my own solid solution, shown above. The yellow atoms are on a regular lattice, and the small purple atoms are also squeezed into the gaps. I have shown both the yellow and purple atoms a bit smaller than they really are, so that you can see the purple atoms, at full size you only see little glimpses of purple.
If you look carefully, you’ll see that the purple atoms aren’t uniformly distributed, they are kind of clustered, there are more in the bottom-left part of the crystal. I don’t know why. Once the crystal has grown around a purple atom it is trapped, it cannot diffuse around as the salt ions can in a liquid solution. The carbon in steel may show the same feature, so I’d like to know why. I’ll have to grow more of these crystals and look carefully at how they grow.