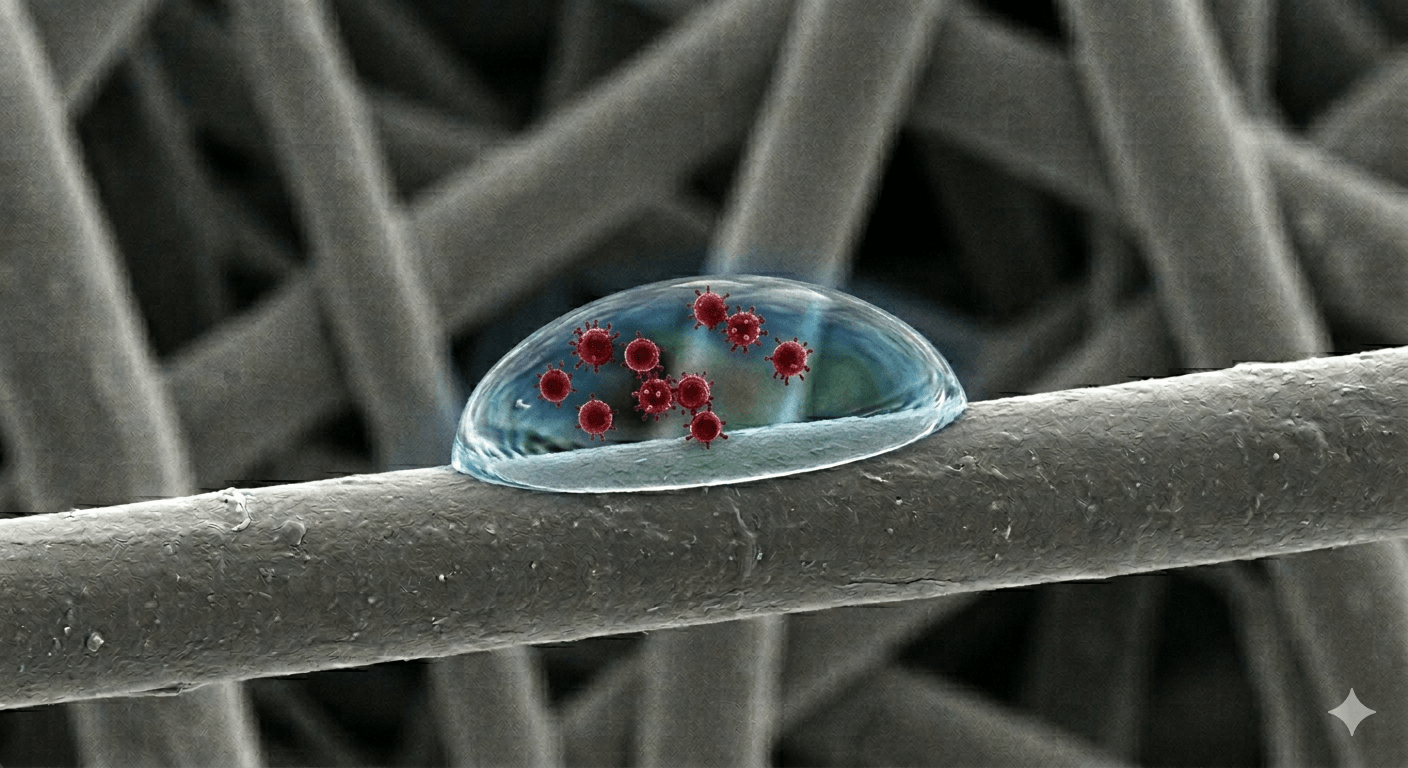
Masks exist to filter out harmful viruses and bacteria from the air the wearer is breathing. This protects the wearer, and if the mask wearer is infected, it protects others in the same room. Masks are air filters worn on the face and air filters are typically meshes of tiny fibres – illustrated in the rather shonky AI image above. They work by the virus-containing droplets in the air sticking to the fibres inside the filter, and so not going through the mask.
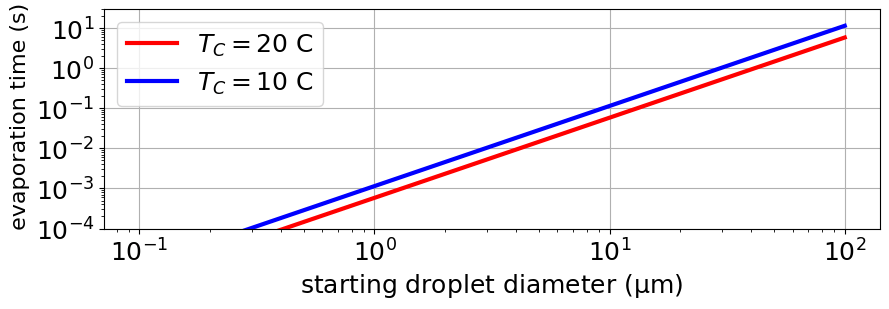
But then what happens to the virus-containing droplets stuck to fibres inside the filter material? There is understandable concern that the mask could be contaminated by these droplets, and that perhaps later infectious virus could be dislodged from the mask and go on to infect someone.