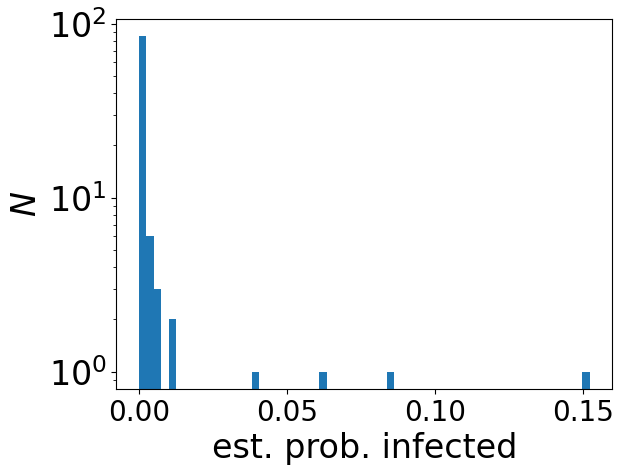
Maybe not. The basic model for the transmission of infectious diseases like COVID assumes that there is such a thing as an infectious dose of a virus, and that these doses act independently. By independently I mean that if you are exposed to one infectious dose, then the probability that you do not become infected is some probability, call it p, while if you are exposed to two infectious doses, then the probability that neither of them infects you is p2 — independent probabilities multiply. As 2 here is an exponent, the probability you avoid infection decreases exponentially with the number of these doses. This predicts a sharp – exponential – increase in the probability of becoming infected, with increasing amounts of virus.
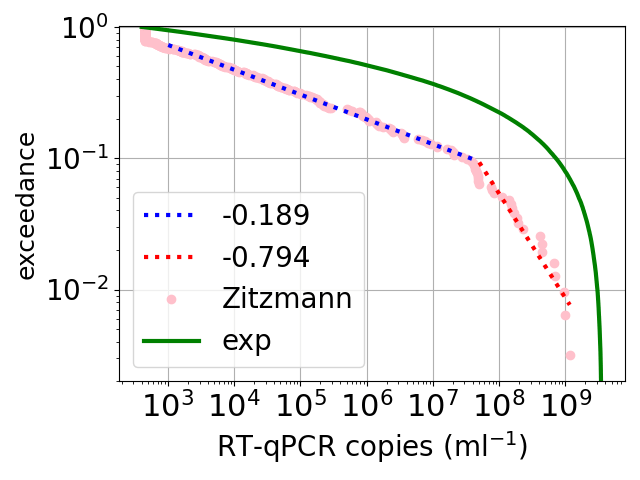
We do not have data on infections in people, but we do have data on infections for cells growing in culture (ie growing in a petri dish or similar). And this exponential variation is not what is observed. See the figure above*. The data are the blue discs and are from the work of Jaafar et al.. The fit of an exponential function is shown as the green dotted curve. The fit is terrible.