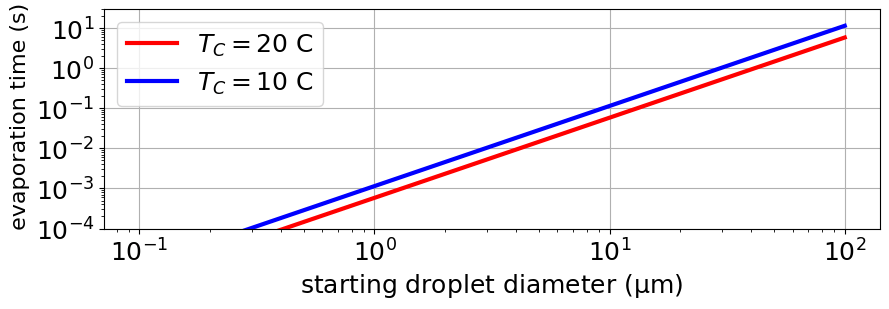
The evaporation of water droplets is a lot more complex than you’d think or hope. One reason is that small water droplets, at least in not very humid air, evaporate so fast that the evaporation cools them. This is just like when you blow on hot coffee to cool it, you increase the evaporation rate and this cools the coffee. Evaporation takes energy, called latent heat levap, which for water is large. At room temperature the latent heat is a bit less than 10−19 J per water molecule that evaporates. Calculations by Hardy et al. find that it can cool an evaporating water droplet by more than 10 C.
Author Archives: Richard Sear
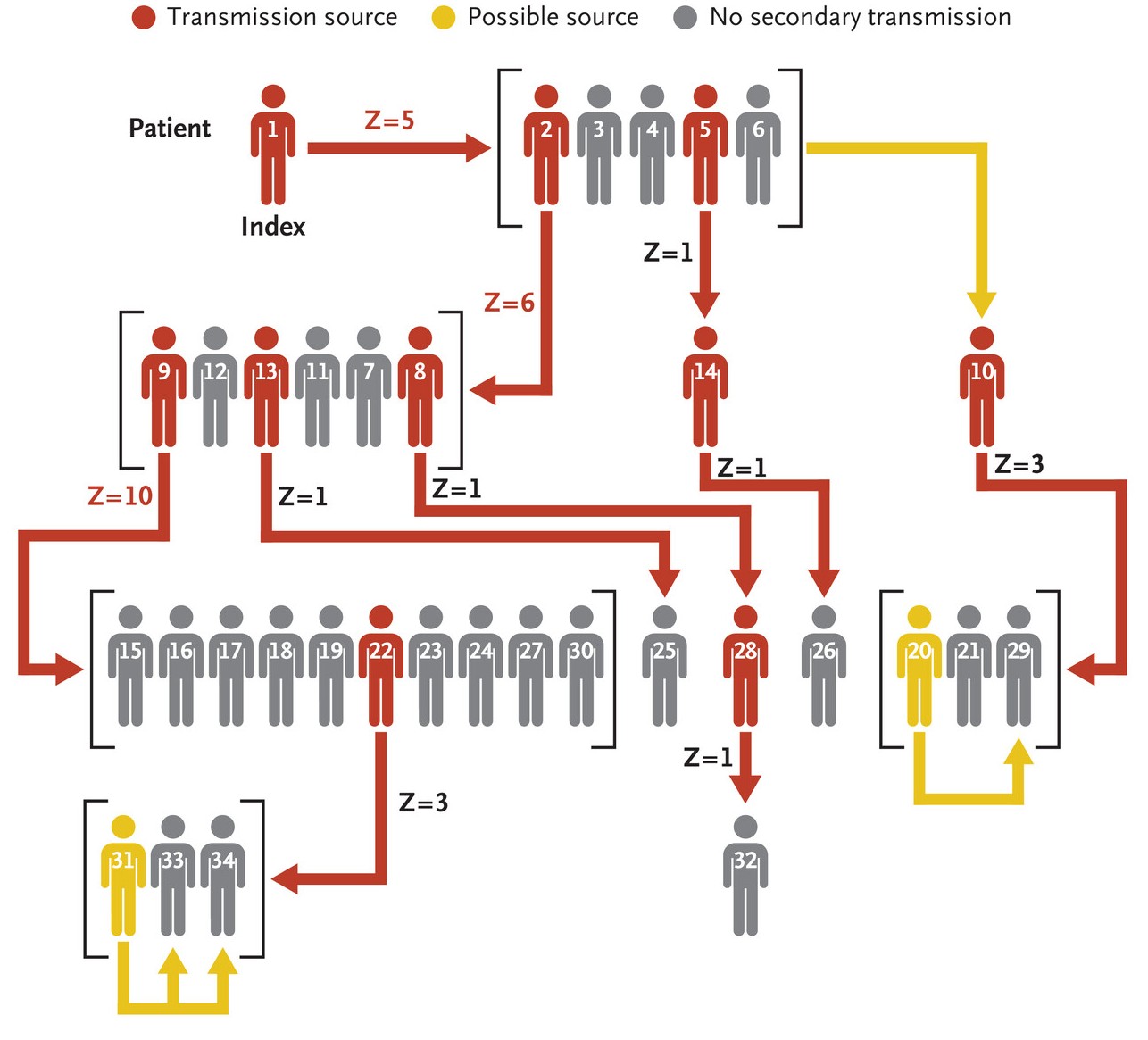
Infectious disease superspreaders on ships
At the moment two ships are in the news for infectious disease outbreaks, the MV Hondius for an outbreak of the Andes strain of hantavirus (ANDV) and the Ambition for a norovirus outbreak. This outbreak on the Handius has resulted in three deaths to date. These are just two of a long line of plague ships, including the Diamond Princess during the early stages of the COVID pandemic. Viewed from one way, this is perhaps unsurprising: large cruise ships squeeze several thousand passengers and crew into a confined space that looks a perfect place to transmit disease from one person to another. But I am bit surprised both that the companies that run these ships don’t seem to be doing anything to stop these regular bursts of bad PR, and that potential passengers are not put off by the risk of boarding a potential plague ship.
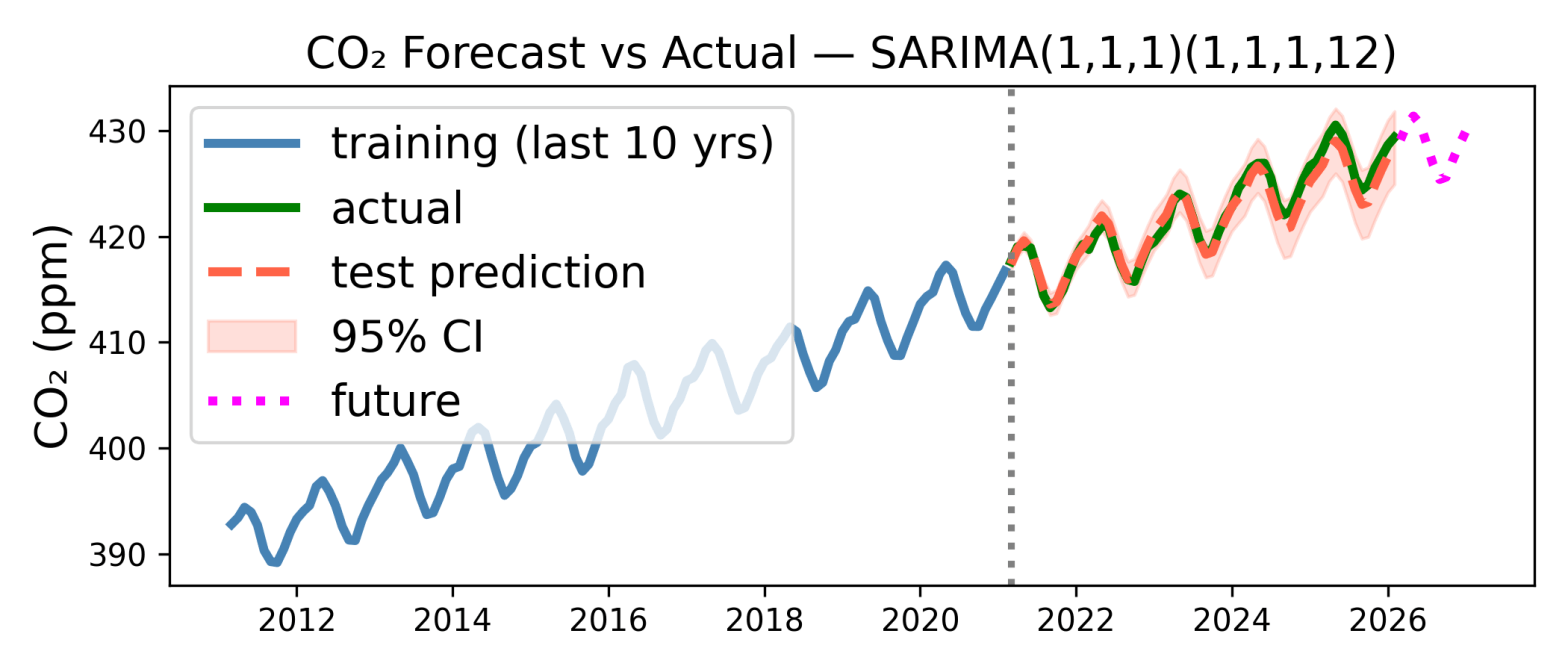
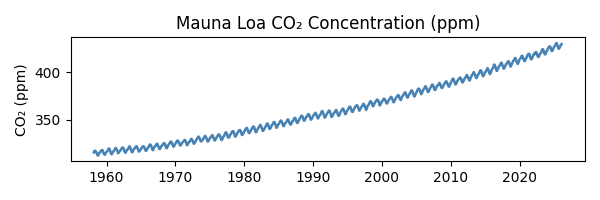
Predicting next year’s CO2 levels with simple machine learning
Due to us burning oil, coal, gas etc etc, the amount of CO2 in Earth’s atmosphere is increasing, and has been for well over a century. This is now well documented. Some of the first measurements were made by Charles David Keeling on the Muana Loa volcano*. He started in March 1958, and the measuring continues to this day. Katherine Bourzac has written a fascinating article about the history of these measurements, which with a couple of interruptions have continued to this day, almost 70 years of data. The data** is here:
Hunting by electric fields, and with the Ampullae of Lorenzini
This beautiful image is of the small-spotted catshark, and is by Hans Hillewaert (Wikimedia). They are carnivores and hunt on the seabed, where I think vision is poor. So they have evolved to hunt, in part, by sensing the electric fields due to their prey. This was shown over 50 years ago, with some beautiful work by AJ Kalmijn. There is a lovely Scientific American article on this, by RD Fields, in 2007.
The difference between insanity and genius is measured only by success and failure
The title is a quote by the manga artist Masashi Kishimoto. It is one of many quotes linking insanity and genius, one of them dating all the way back to Aristotle. But now, finally, we can quantify how similar insanity and madness, it is: 0.5810. This may not look that high but insanity is closer to madness than sanity, sanity and madness only have a similarity of 0.4114.
Apps for understanding better the air we breathe
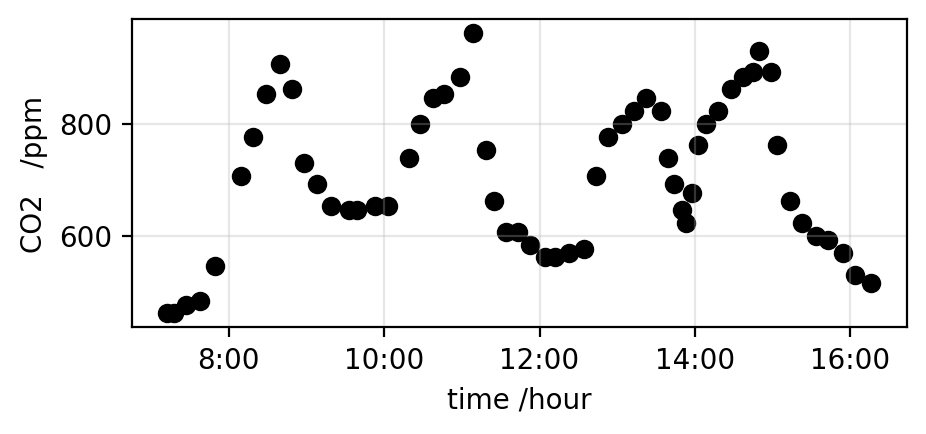
I have been suckered* into running a schools event next week, so spent most of the day writing an app to allow school kids to plot the CO2 concentration of indoor air, and do some very basic analysis. The app is now running, both on Streamlit Community Cloud and Railway*. It takes csv files that have some fraction of a day’s data on CO2 concentration in the air, and plots the data. A plot from the app is shown above. The data on CO2 levels is from a Canadian school, I thought some data on a school might be good to interest the school children.
Rattlesnakes and detecting warmth a thousandth of a degree at a time
This* is the head of a western diamond rattlesnake, a species of pit viper that is common in the USA and Mexico. Pit vipers are a family of species of snakes that called pit vipers because they have a specialised sensor organ – a pit – to detect thermal radiation (ak infrared or IR radiation). This in addition of course to their eyes which detect visible radiation. If you look carefully you can see a pit in the picture above. From left to right it lies about halfway across between the eye and the nostril, and it is close to the bottom of the head. It is hard to see because it is a pit so you really just see the shadow.
Some are born geometers, some achieve competence at geometry, and some have geometry thrust upon them
When fluids, such as air and water, flow slowly and in small systems, the equations that govern the flow simplify and become the Stokes equations. These are linear and so in simple geometries can be solved analytically, with pencil and paper. And so even before the advent of computers, a number of solutions were obtained. One of them was for flow through a circular hole in a (thin) plate. This was solved in 1891 by one Ralph Allen Sampson. The paper is a bit of a horror story of fancy maths* (in it he did many other things other than the flow through the hole) but unless I am mistaken* his result for the flow through the hole was amazingly elegant and simple, at least when viewed the right way.
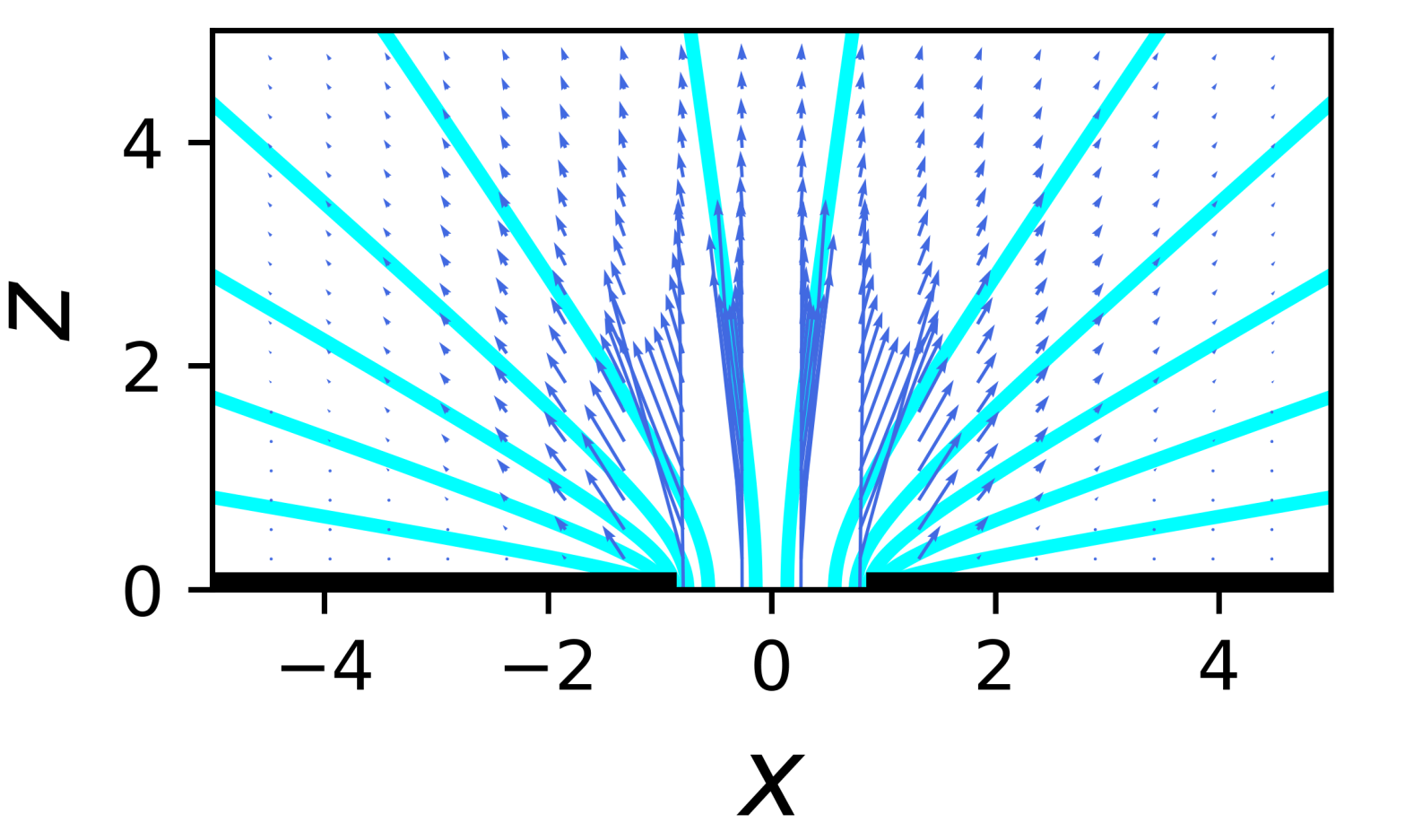
Pushing on fluids at points far from, and very near, walls
This is just a post on me teaching myself a bit about the flow patterns that occur when you apply a small force at a point in a fluid. Small here means that the flows are sufficiently small and slow that the flow is Stokes flow, i.e., the Reynolds number is close to 0.
I used LLMs a fait bit, mainly MS Copilot. The LLMs helped with elements of the problem I am interested in, but could not do the whole thing. The code to work through the flow patterns is on GitHub if you’re interested. In that sense LLMs are – very roughly speaking! – currently at the level of say a final year undergraduate lecture course on Stokes flow, but no higher. Perhaps because their training sets contains a few fluid mechanics textbooks but not enough examples beyond that. To be fair on Copilot etc clear descriptions of the problem are hard to find in the literature …
Surely microplastics are a very small problem?
I quite like Wired magazine but it has run an article called: People Who Drink Bottled Water on a Daily Basis Ingest 90,000 More Microplastic Particles Each Year. I just cannot make sense of this. In particular I think the number 90,000 is supposed to be scarily big. But 90,000 particles of this size in one year is an absolutely tiny amount. Let me try and put the figure of 90,000 in context.