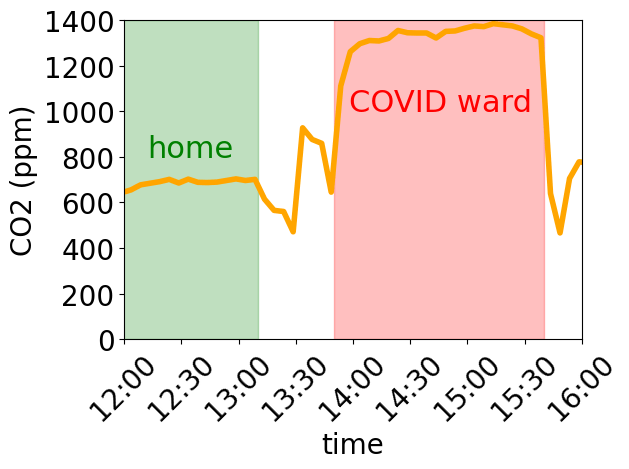
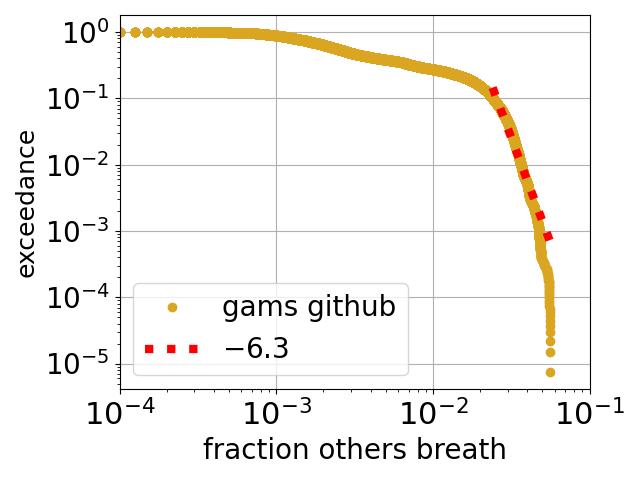
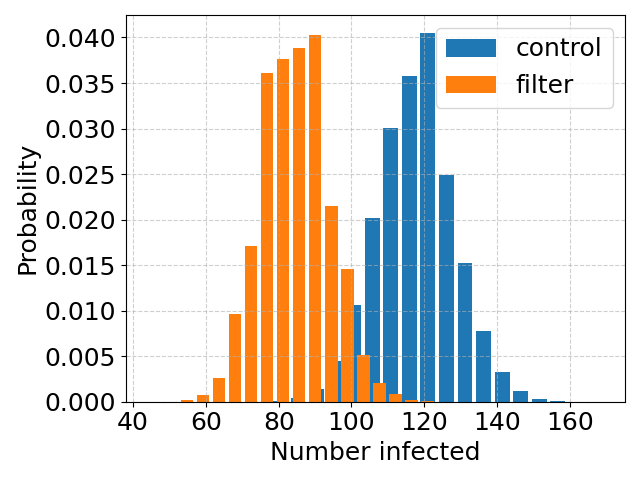
The one formula that I use above all others in my research is that for the diffusion constant of a dilute species in a fluid, called the Stokes-Einstein (or sometimes Stokes-Einstein-Sutherland) equation:
DSE = kT/(6 π η RH)
The diffusion constant DSE of something with a size (hydrodynamic radius) RH is just the thermal energy kT divided by 6π times the product of the viscosity of the fluid, η, and the hydrodynamic radius, RH. All it tells you is that large species in viscous fluids diffuse more slowly than small species in less viscous fluids.