The title seems a reasonable question to ask. If you are pretty close to someone and talking to them, then you are facing them and so more-or-less in the direct path of their exhaled breath. A single exhaled breath contains very roughly of order a few hundred aerosol droplets* (typically all of which are so small they are invisible). Depending on how close you are to someone you are talking to, the air you are inhaling may contain say 1 to 10% air they have breathed out. At a breath every few seconds then during a chat with someone you could inhale anywhere between tens and many thousands of tiny droplets that they have exhaled – a perhaps unattractive thought but this is what happens when we share the air with fellow humans. Something, we as social creatures, do a lot.
Author Archives: Richard Sear
Does an infectious dose of a virus exist?
Maybe not. The basic model for the transmission of infectious diseases like COVID assumes that there is such a thing as an infectious dose of a virus, and that these doses act independently. By independently I mean that if you are exposed to one infectious dose, then the probability that you do not become infected is some probability, call it p, while if you are exposed to two infectious doses, then the probability that neither of them infects you is p2 — independent probabilities multiply. As 2 here is an exponent, the probability you avoid infection decreases exponentially with the number of these doses. This predicts a sharp – exponential – increase in the probability of becoming infected, with increasing amounts of virus.
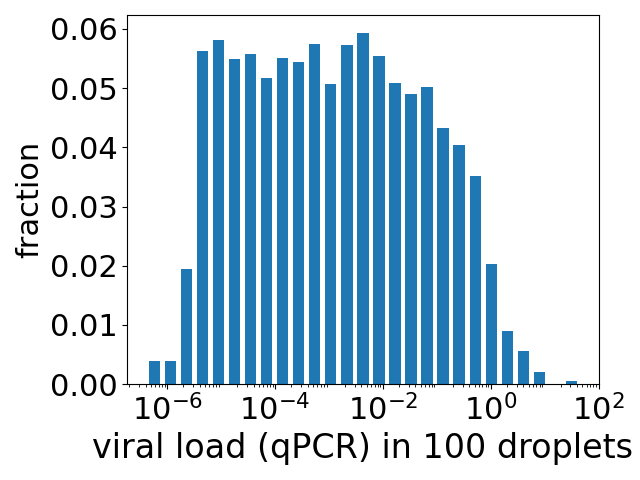
We do not have data on infections in people, but we do have data on infections for cells growing in culture (ie growing in a petri dish or similar). And this exponential variation is not what is observed. See the figure above*. The data are the blue discs and are from the work of Jaafar et al.. The fit of an exponential function is shown as the green dotted curve. The fit is terrible.
The Welsh ambulance service declares a “critical incident” and still the NHS in Wales does not “follow the science”
A couple of days ago – 30th December 2024 – the Welsh ambulance service declared a critical incident. According to the Wales Online report, the head of the ambulance service, Stephen Sheldon said
“It is very rare that we declare a critical incident, but with significant demand on our service and more than 90 ambulances waiting to hand over patients outside of hospital, our ability to help patients has been impacted.”
Welsh (and English) hospitals are under great pressure from a “quad-demic” of flu, RSV, COVID and norovirus. Three of these diseases are transmitted across the air: flu, RSV and COVID. The fourth: norovirus, is food and waterborne. This pressure has prompted the introduction of both visiting restrictions and masking requirements in Welsh hospitals.
The visiting restrictions for Swansea’s hospitals near where my mother lives are here. If you follow the link you will see a large picture of surgical masks, so looks like visitors to hospitals will be handed surgical masks, to try and reduce the spread of flu (and RSV, COVID etc).
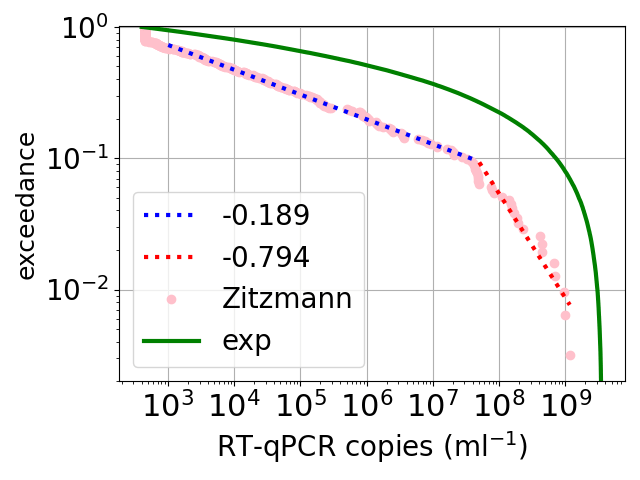
Sampling from exponential growth of viruses
The plot of the distribution of SARS-CoV-2 viral loads, in infected people. SARS-CoV-2 is the virus that causes COVID. The x axis is the estimated number of viruses per millilitre of mucus, in a sample taken from an infected person. The y axis is the exceedance, which is the fraction of the samples that have a viral load larger than the number along the y axis. So for example, for a viral load around 3 * 107 virus/ml, the exceedance is 0.1, so 10% of the patients had more than 3 * 107 virus/ml. Note that the range of viral loads spans a range of over a million.
100 men walk into a badly ventilated bar, how many catch COVID?
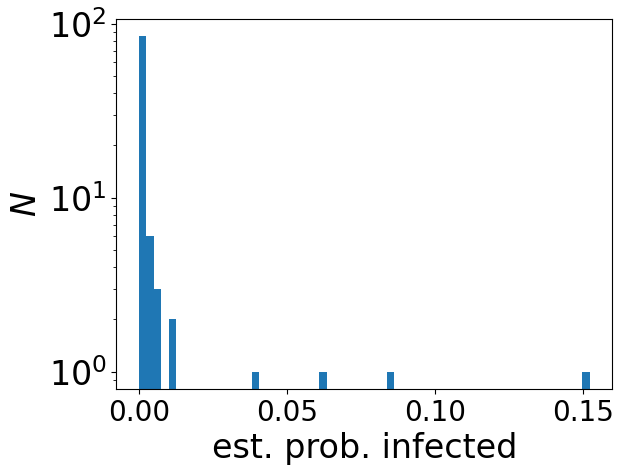
Early on in the pandemic, the aerosol scientist Jose-Luis Jimenez made available a Google Sheets implementation of the standard model for airborne disease transmission: the Wells-Riley model. This was a very nice idea, to provide a rational way to estimate the risk of transmission. The Sheets implementation is very open about the (many) assumptions made by the model, and it cites supporting literature. It is a good piece of work.
Is mpox mainly a sexually transmitted disease with rarer transmission across the air like COVID?
The latest virus to make the news is the Clade Ib strain of mpox, it looks like a person was infected overseas but infected two members of their household in the UK. There seems to be a lot of confusion as to how it spreads, The Guardian, the NHS, and the USA’s CDC all have different ideas.
The COVID-19 enquiry is back and we have weak leadership, responsibility dodging and lack of the full range of technical expertise
The UK’s COVID-19 enquiry is back and it does not make cheerful watching. I first watched testimony of Dr Barry Jones, Chair of the Covid Airborne Transmission Alliance (CATA). He is was impressive, and is clearly very very unhappy with the IPC Cell (IPC=Infection Prevention and Control), and I agree he has reason to be. The IPC cell was set up at the start of the pandemic and is somehow part of the then Public Health England (PHE) (now renamed as Health Security Agency) and the NHS. It issued “guidance” on how to prevent the spread of COVID-19, that went to PHE who seemed to rubber stamp it and send it to hospitals who all obeyed it. I then watched testimony (continued here) from the Chair of the IPC, for a year of the pandemic, Dr Lisa Ritchie. Dr Ritchie was less impressive.
Move over quantum computing, neuromorphic iontronics is here
About 100 years ago, quantum physics was invented. Then around 70 years later in the ’90s along came quantum computing. And now quantum computing is quite topical. Similarly, over a hundred years ago electrokinetics was developed, then in the early 2000s, along came iontronics, which is on the rise. Iontronics is to electrokinetics roughly as quantum computing is to quantum physics. Quantum computing is an application of quantum physics to doing computations, or making machines to do computing. Electrokinetics is moving around ions etc, often in solution, while iontronics is moving around ions to do computations, or make a computer.
Bullshit images
Hicks, Humphries and Slater have recently published a paper entitled ChatGPT is bullshit. First of all, 10/10 to them for the paper title. Their point is simple:
Applications of these [LLMs like ChatGPT] systems have been plagued by persistent inaccuracies in their output; these are often called “AI hallucinations”. We argue that these falsehoods, and the overall activity of large language models, is better understood as bullshit in the sense explored by Frankfurt (On Bullshit, Princeton, 2005): the models are in an important way indifferent to the truth of their outputs.
I think this is a fair point, LLMs do produce results that are just wrong, and seem rather indifferent to whether their output is correct.
Not all viruses are the same
Above are schematics of 4 very different viruses (schematic from the PDB). From left to right: The giant of the 4 is the mpox or monkeypox virus, that is a current cause of concern, the WHO declared mpox a “public health emergency of international concern” last week. We appear to have a very poor understanding of how mpox is transmitted except via “close [(broken?) skin to (broken?) skin, prolonged?] contact”. Next is HIV, which is transmitted via sex and blood. The third is SARS-CoV-2, the cause of COVID-19 and the recent pandemic, which is mainly transmitted across the air. Finally, the tiny one is poliovirus, the cause of polio, which is transmitted in food/water.