 On Thursday I was quietly working in my office when a professor I don’t know, from Brunel University, phoned me. He is an organiser of next week’s 4th International Conference on Advances in Solidification Processes – a conference on metal crystallisation. One of their plenary speakers had bailed on them. He asked me to step in, so I said yes.
On Thursday I was quietly working in my office when a professor I don’t know, from Brunel University, phoned me. He is an organiser of next week’s 4th International Conference on Advances in Solidification Processes – a conference on metal crystallisation. One of their plenary speakers had bailed on them. He asked me to step in, so I said yes.
So I have hurriedly set about writing a talk that metals science guys will hopefully find interesting. I don’t work on metals myself. I do work on crystallisation but mostly of molecules from solution. But I have been reanalysing some old (60 years!) data on the freezing of small droplets of liquid tin.
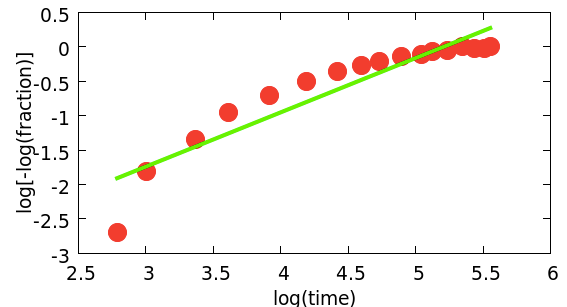
However, there is a slight problem. My best guess at a model predicts that data for the fraction of droplets that still liquid, i.e., have not frozen, as a function of time, should fall on a straight line on what is called a Weibull plot*. The plot is above. The red circles are data for the freezing of droplets of tin.
Obviously, the red circles do not fall on a straight line. The green line is the best straight line fit and it is clearly a terrible fit. What to do?
When a model does not fit, this is telling you that (at least) one of the assumptions of the model are wrong. Here, it may be the assumption that each tin droplet has the same number of sites where freezing can start. That may be something for the metals guys to think about. Their standard theory predicts not only that the data should be on a straight line but that this line should have a slope of one. We both have to work to do to understand what is going on here.
Bur before that I had better brush up on knowledge of metals and get cracking on writing on my talk.
* By definition, on a Weibull plot, the y axis is log[-log(fraction of droplets still unfrozen)] and the x axis is log(time).